Rules on Handling Genome-edited Crops, Food Products and Feeds in Japan
Summary
・Use of any living modified organism is strictly regulated by domestic acts for each purpose.
・Organisms obtained through genome-editing technologies may be regulated by the acts, based on the type of genome-editing techniques and processes used.
・When using genome-edited organisms that are not regarded as living modified organisms, users should consult and submit information regarding the organisms to the government for information disclosure.
The handling of genome-edited organisms has been discussed within the framework of living modified organisms worldwide; organisms are often genetically modified in the process of genome-editing. In Japan, the Cabinet approved the Integrated Innovation Strategy (Cabinet Office) in June 2018. Clarifications regarding the handling of genome-edited organisms under the Act on the Conservation and Sustainable Use of Biological Diversity through Regulations on the Use of Living Modified Organisms (Cartagena Act” hereafter; the domestic law for assessing the influences on biodiversity) and the handling of genome-edited food products and additives under the Food Sanitation Act were requested to be provided by the end of the 2018 fiscal year.
On February 8, 2019, the Ministry of the Environment (MOE) issued a report titled ‘About the Handling of Organisms Produced by the Use of Genome Editing Technology that Do Not Match the Definition of “Genetically Modified Organisms” under the Cartagena Act’ (MOE 2019). On March 27, the Ministry of Health, Labour and Welfare (MHLW) issued a report titled ‘Handling of Foods Obtained Using Genome Editing Technology in Terms of Food Hygiene’ (MHLW 2019a). They collected public comments on the details of the notification and reported the handling procedure on September 19 (MHLW 2019b). The Ministry of Education, Culture, Sports, Science and Technology (for academic use) and the Ministry of Economy, Trade and Industry (for bio-industrial use) issued their handling policies (MEXT 2019, METI 2019). The Ministry of Agriculture, Forestry and Fisheries (MAFF) collected public comments on the handling policies and presented specific procedures on October 9 (MAFF 2019). The MAFF also issued the ‘Feed Safety Guidelines for Genome Edited Feeds and Feed Additives’ (MAFF 2020) on February 7, 2020.
Terms necessary for discussing regulations of products developed by genome-editing technology
In this section, the terms necessary for discussing genome-editing technology are explained first. Then, the rules for handling genome-edited organisms in Japan are explained from the perspectives of influences on biodiversity and food and feed safety.
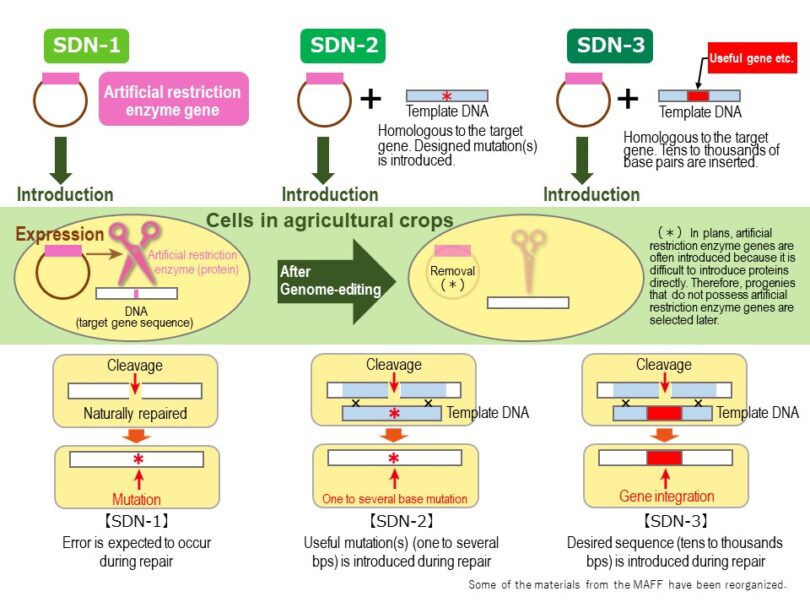
SDN (Site-Directed Nuclease): division of genome-editing techniques (SDN-1, SDN-2, and SDN-3; Fig. 1). The SDN-1 technique induces the deletion, insertion, or base substitution of nucleotides at the target site. In SDN-1, the target DNA sequence is cleaved by CRISPR/Cas9, etc. and naturally repaired. The process of mutagenesis is the same as natural mutations: it involves failures in DNA repair. The SDN-2 technique involves the introduction of DNA fragments prepared extracellularly (“foreign DNA fragments” hereafter) to the target gene. The foreign DNA fragments are basically the same as the target sequence but contain one or more mutations (deletion, insertion, or base substitution). After the target site is cleaved, the designed mutation is introduced to the target gene by means of homology directed DNA repair using a mutated template. The mechanism of SDN-3 technique is similar to that of SDN-2. With the SDN-3 technique, tens to thousands of base pairs of foreign DNA fragments (e.g., an entire gene) can be integrated into the genome.
Null-segregant: A living organism from which the genome-editor genes, once integrated into the genome, have been removed by crossing, etc. The genomes of animals and microorganisms can be edited through the direct introduction of genome-editing tools (protein for TALEN, protein-RNA complex for CRISPR/Cas9) into cells. However, the direct introduction of the tools into plant cells is a challenging process. The tool genes are often introduced and expressed by conventional gene recombination methods, and thus the host becomes a genetically modified plant. Upon the completion of genome-editing, the tool genes can be removed by repeated crossing, etc.
Off-target: unexpected mutation that occurs at a non-target site. In the genome-editing technology, the introduction of mutations into non-targeted sequence occurs less frequently compared to conventional mutagenesis techniques. Still, a sequence similar to the target may be mistakenly recognized and cleaved.
Handling in the Cartagena Act
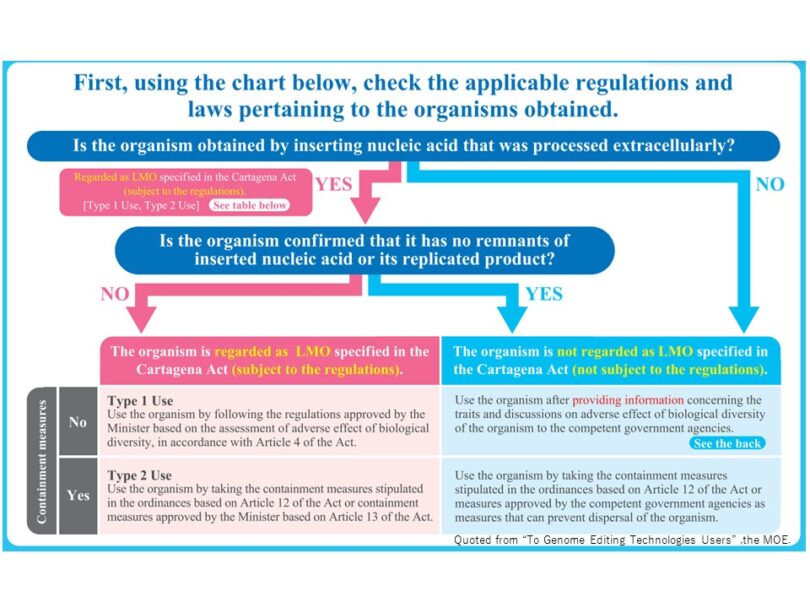 Fig.2: Policies on the classification and handling of genome-edited organisms under the Cartagena Act.
Fig.2: Policies on the classification and handling of genome-edited organisms under the Cartagena Act.
Living organisms regulated by the Cartagena Act are defined in Article 2. In this act, a “living modified organism” is defined as “an organism that possesses nucleic acid, or a replicated product thereof, obtained through use of the technologies for the processing of nucleic acid extracellularly.” The key factor in examining the handling of genome-edited organisms under the Cartagena Act is whether the organism falls into a “living modified organism,” i.e., whether it contains a foreign gene or a foreign DNA/RNA fragment (“foreign gene” hereafter). Therefore, it is first assessed whether a foreign gene is introduced into the organism in the process of genome-editing (Fig. 2).
(1) If a foreign gene is not introduced, the organism is classified as an organism that is not subject to the regulations. e.g., genome-edited organisms produced by delivering artificial nuclease proteins (TALEN, etc.).
(2) If a foreign gene is introduced, the organism is not subject to the regulations upon confirming the removal of the gene. e.g., Genome-edited organisms produced by delivering protein/RNA complexes (CRISPR/cas9 etc.) directly to the cells or by transferring and expressing the tool genes transiently in the cells. The tool genes must not remain in the cells eventually. Null segregants from which integrated foreign genes are ultimately removed also fall under this category.
Organisms that continue to contain a foreign gene or for which the removal of the gene cannot be guaranteed are regulated as living modified organisms.
If the organism is edited using the SDN-1 technique and falls into either case (1) or (2), it may not be regulated by the Cartagena Act (Fig. 3). However, organisms that are edited using the SDN-2 or SDN-3 technique are regarded as living modified organisms, as they obtain foreign sequences independently of the editing processes.
The policies pertaining to the handling of genome-edited organisms that are not considered “living modified organisms” under the Cartagena Act are not legally binding. However, those who use such organisms are strongly requested to submit information on the characteristics of the organisms and their potential adverse impact on biological diversity, etc. (Table 1) to the government agency to which the competent minister belongs. Once the information is accepted, a submission is not necessary for every use unless the information changes in some way (e.g., through further genetic modification). A submission is not necessary if the organisms are used in accordance with “Type 2 Use in Cartagena Act” in closed environments.
Table 1. Information requested from the developers by the MOE
(a) The fact that the organism does not possess remnants of extracellularly processed nucleic acid or its replicated product, as stipulated in the Cartagena Act (including bases of decision)
(b) Taxonomical species of the modified organism
(c) Method of genome editing used for the modification
(d) Modified gene and its functions
(e) Change of traits added by the modification
(f) Whether there is a change of traits other than those described in (e) . Please describe the changes, if any.
(g) Usage of the organism
(h) Discussion on possible influences on biological diversity when the organism is used.
Handling under the Food Sanitation Act
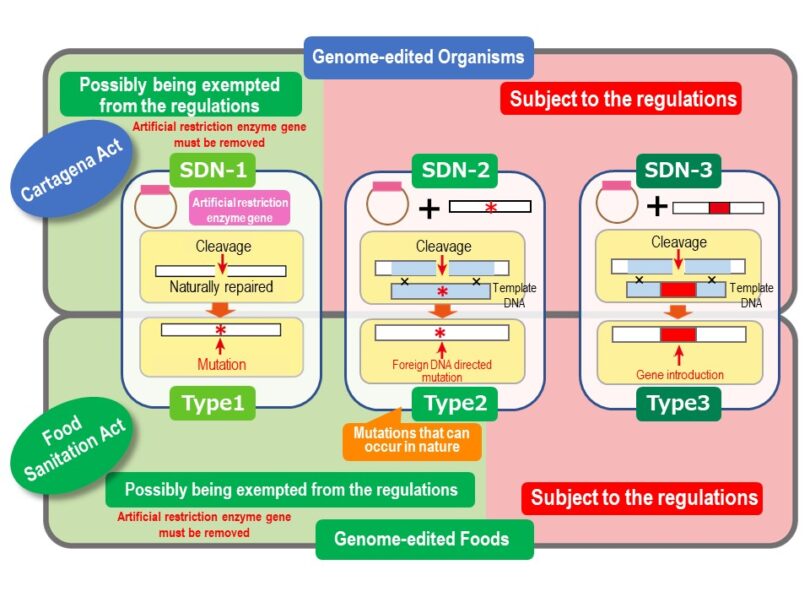 Fig.3: Comparison of the handling procedures under the Cartagena Act (living organisms) and the Food Sanitation Act (food products).
Fig.3: Comparison of the handling procedures under the Cartagena Act (living organisms) and the Food Sanitation Act (food products).
Under the Food Sanitation Act, genome-editing technologies are classified into either Type 1, Type 2, or Type 3, which employ SDN-1, SDN-2 and SDN-3 organisms, respectively. Independent of their class, genome-edited foods in which the tool genes remain (or their removal is not guaranteed) are subject to the Act for safety inspection. As for the tool gene-free organism, the judgment on whether the organism is subject to the regulations as a living modified organism is slightly different from that specified under the Cartagena Act (Fig. 3).
Foods classified as Type 1 may be exempted from the regulations of the Food Sanitation Act. Those classified as Type 3, which contain a foreign gene, are subject to the regulations. The judgements made by the Cartagena Act and the Food Sanitation Act may differ with respect to Type 2. The Type 2 technique involves the introduction of a foreign DNA fragment to mutate several bases in the target sequence. Although a foreign DNA segment is not introduced in Type 1 (SDN-1), it can sometimes induce mutations at higher magnitude than Type 2 (SDN-2). The report by the Research Committee for Newly Developed Food stated: “… Deletion and substitution of bases due to repair of cleavage sites of artificial restriction enzymes, deletion of genes that may occur in nature, and insertion of mutations of one to several bases as a result do not fall under the recombinant DNA technology under the Food Sanitation Act …,”. Therefore, it was decided that the assessment is carried out on the basis of the final product (product-based). However, the kind of Type 2 products that are exempted from the regulations have not been decided. It is likely to be examined when actual information is submitted.
It was decided that the handling of additives manufactured using genome-edited organisms should be treated at the same level (or a more relaxed level) than that for foods. On the other hand, the submission of necessary information, including the characteristics specific to each additive, is requested. This conclusion is based on the premise that the compositional standards of additives are officially established in general. If the technology used does not correspond to recombinant DNA technology, the submission of information is requested as in the case for the handling of foods. Submission is not requested for highly purified additives.
Information regarding genome-edited foods that are not subject to the regulations of recombinant DNA technology must be submitted to the Consumer Affairs Agency (CAA) before it is launched in the market (on a voluntary basis and no penalty is involved for violation). The ministry states that the accumulation of data related to genome-edited foods is socially important, and that the government needs to address the concerns of consumers on new breeding technologies. Table 2 presents the information that must be submitted. For item C, genomic sequences presumed to have high probability of off-target editing need to be predicted using multiple search tools. If a mutation is introduced unexpectedly, a confirmation must be issued that the mutation does not result in the production of allergens or enhancement of known toxic substances.
Table 2. Information requested from the developers by the CAA
(a) Name of item and breed and summary (usage and intended use) of the developed food
(b) Method of genome editing technology used and details of modification
(c) Information on confirmation that there are no remaining foreign genes or their parts
(d) Information on confirmation that confirmed changes in DNA do not cause production of new allergens having adverse effects on human health or increase of known toxic substances contained
(e) For items in which modification affecting the metabolic system was performed in order to increase or decrease specific components, information on changes in major components (nutrient components only) related to the target metabolic system
(f) Year and month of marketing (Notify the CAA of it after marketing)
Developers/users can ask the CAA for a consultation prior to the submission of information. It seems important to consult with the authority on whether an organism is subject to the regulations and on how to prove that it is foreign gene-free. If a foreign gene is found in a genome-edited food product, etc. (that has already been marketed as a product not applicable to recombinant DNA technology), it will be considered to have been distributed without authorization. Such an incident will lead to loss of trust in the developer as well as the management authority. Information sharing between developers/users and the ministry can prevent the unauthorized distribution of genetically modified foods. Thus, although not obligatory, both prior consultation and the submission of information are strongly requested by the regulator.
Handling under the Feed Safety Act
Feed crops and feed additives produced using genome-editing technologies are assessed under the Feed Safety Act. If the final products contain foreign genes or fragments of such genes, they are regarded as recombinant DNA technology derivatives. Such items are subject to a safety assessment as per the Procedures for Safety Assessment of Feeds and Feed Additives Derived from Recombinant DNA technology (MAFF Notification No. 1780 of 2002). For other items produced by genome-editing, their need for submission and safety assessment will be determined on a specific case-by-case basis by the MAFF. Table 3 presents the information regarding genome-edited feeds and feed additives that must be submitted by the developer, etc. to the MAFF.
Table 3. Information requested from the developers by the MAFF
(a) Name of item/breed and summary (usage and intended use) of the developed genome edited feed
(b) Information on method of genome editing technology and genetic modification used
(c) Information on confirmation that there are no remaining foreign genes or their parts
(d) Information on confirmation that confirmed changes in DNA do not cause increase of known toxic substances
(e) Information on changes in major components (nutrient components only) related to the target metabolic system for items which modification affecting the metabolic system was performed in order to increase or decrease specific components
(f) Year and month of marketing (Notify the MAFF of it after marketing)
Accepted information regarding genome-edited foods, food additives, feeds, and feed additives are disclosed as a notification without infringing the applicant’s intellectual property.
Reference (all checked on October 10, 2025)
Ministry of the Environment 2019. https://www.env.go.jp/press/106439.html (Japanese)
Ministry of Economy, Trade and Industry 2019. https://www.meti.go.jp/policy/mono_info_service/mono/bio/cartagena/detailed_info/genome.html (Japanese)
Ministry of Health, Labour and Welfare 2019a. https://www.mhlw.go.jp/content/11121000/000494346.pdf (Japanese)
Ministry of Health, Labour and Welfare 2019b. https://www.mhlw.go.jp/content/000550824.pdf
Ministry of Agriculture, Forestry and Fisheries 2019. https://www.maff.go.jp/j/syouan/nouan/carta/tetuduki/attach/pdf/nbt_tetuzuki-8.pdf (Japanese)
Ministry of Agriculture, Forestry and Fisheries 2020. https://www.maff.go.jp/j/syouan/tikusui/siryo/attach/pdf/ge_todokede-9.pdf
Ministry of Education, Culture, Sports, Science and Technology 2019. https://www.mext.go.jp/lifescience/bioethics/files/pdf/n2189.pdf (Japanese)
Consumer Affairs Agency 2019. https://www.caa.go.jp/policies/policy/food_labeling/quality/genome/ (Japanese)